Evidence-based probiotics, the gut microbiome and immunity – the scientific and holistic way forward

In recent peer reviewed papers from the international biomedical research journals, the gut microbiome is now seen clearly to be a major instigator of immune health, not only for the gut but also for lung health (Ref 1,2).There are more cells in the body’s microbiome than there are somatic cells in the body. There is more DNA within cells of the gut microbiome directing immunity than there is in the entire somatic cell count of the body. An optimised gut microbiome will have a higher preponderance of commensal organisms if acted upon directly by a proven and quality probiotic (e.g. Provita Protect – the only veterinary probiotic whose claims and proven efficacy have been accepted and authorised by the VMD). This probiotic-enhanced gut microbiome with its higher proportion of commensal organisms in turn will produce higher amounts of SCFA (short chain fatty acids)from the commensals which are now known to be the signalling mechanism from the gut to the body’s immune systems and especially to the immune system of the lungs. “Cross Talk” between the gut and the lungs is now a proven scientific concept and is the “buzz word” in modern immunology. This new concept explains not only the healthier gut and lower incidence of scours seen with a licensed probiotic such as Provita Protect, but also explains the significantly lowered incidence of calf respiratory disease which is also seen as a bonus when calves receive Provita Protect as has been evidenced by farmers who keep repeatedly using the product for this dual effect. See Table 1 below
| Arrival Treatment | Total | Number of calves
“healthy throughout” |
Calves treated for Scours & Pneumonia |
Percentage calves treated |
| Control | 40 | 21 | 19 | 49% |
| Provita Protect | 55 | 49 | 6 | 11% |
| Provita Protect | 54 | 43 | 11 | 20% |
| Total | 149 | 113 | 36 |
Table 1 from Provita Protect Drug Master File Submission for VMD Registration-Clinical Trials Section
When is a Probiotic not a Probiotic.?
The resurgence of interest by veterinary surgeons in proven and effective probiotics as alternatives to antibiotics, has been triggered by a number of recent issues:
(1) The global increase in AMR in food animals and its likely transference to humans
(2) The worldwide push back on the use of antibiotics in animal husbandry
(3) New EU regulations on prohibition of prophylactic and metaphylactic use of antibiotics
(4) The realisation that prevention is better than cure and that avoidance of disease is better than treating it, both financially and on welfare grounds.
(5) The overwhelming recent scientific evidence on the importance of high immunity in contributing to a healthy animal and to the realisation of its genetic potential in growth and productivity
(6) The recent published evidence of the multiple and widespread immunological effects conferred by a healthy gut microbiome.
The term ‘Probiotic’
The term probiotic has confused people and a number of veterinarians still regard such substances with a degree of scepticism tending to underestimate the scientific potency of proven probiotic products. Why is this? Well, the answer is relatively simple.
There have been very many unproven and substandard ‘probiotics’ on the veterinary market for quite a number of years now. The confused situation has arisen because in the early days, probiotics were defined as “live microorganisms which when administered in adequate amounts could confer a health benefit on the host”. In other words, it was a ‘possible effect’ rather than a proven effect’. Furthermore, quality control, characterisation of microorganism strains, shelf life of product, number of strains, and regulatory approval of manufacturing premises were not required at that time.
Hence, unproven, unsubstantiated, and unreliable products remained as the majority of probiotic products on the market and were easily accessible by farmers and vets. The lack of standardisation and the lack of proof of any kind of efficacy for the product made it extremely difficult for the veterinarian to distinguish between what was a scientifically evidence based effective compound and what was a totally useless product.
A number of companies do not identify the types and numbers of viable organisms in their products at the time of use, nor have they any clinical trials performed to validate and prove their product. This possibly leads people into thinking that their products have actually been clinically tested and proven to confer specific health benefits when, in fact, no such evidence exists. The end result is the lack of credibility for the field of probiotics as a whole and uncertainty as to which products may benefit animal patients.
The Current Recommendations
Recently groups of specialist international scientists in the microbiome and probiotic fields, mainly in the human sector, have been convening to review and discuss the above issues regarding the lack of international standardisation of the term ‘probiotic’. They have identified the problems and they have set out new criteria in terms of quality controls ,proof of efficacy, and when the term ‘probiotic product’ can be properly used .Their findings were recently published in Frontiers of Microbiology (July 2020) (ref 1).
Scientists have set out criteria that attempt to clarify an established industry definition of probiotics, as the team addresses the term’s misuse, which contributes to confusion about probiotic health benefits.
“The term ‘probiotic’ poses a scientific problem,” says the International Scientific Association for Probiotics and Prebiotics (ISAPP), one of the organisations to take part in the review. ‘It does not distinguish between bacterial strains that have a possible health benefit and strains that have a demonstrated health benefit, as shown in studies.’
The review also calls for probiotics to be supported by at least one clinical trial preferably followed by confirmatory trial(s).
To correctly use the term ‘probiotic’ to describe such strains, the review states that the organism must be identified at strain level and shown to express the relevant trait. Proper characterisation of the strains, shelf life, viable organisms and count, and proof of efficacy of the actual product are necessary if the term probiotic is to be used.
Provita Protect – How is it Different?
Provita Protect is the only veterinary probiotic to be authorised and licensed by the VMD. Copious clinical trial data, veterinary expert reports, and proof of efficacy were submitted in large Masterfile dossiers to the regulatory authorities, in order to acquire this valued authorisation. In other words, Provita Protect uniquely is a proven animal probiotic product, and is thus clearly set apart from most other probiotics in the veterinary marketplace.
Amongst the key finding in the field trials with Provita Protect in young calves were the following:
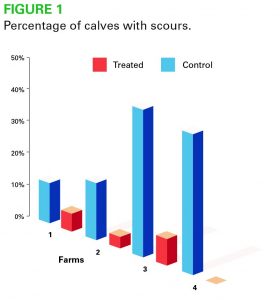
- Less incidence of scours in calves treated with Provita Protect (Fig 1)
- Less incidence of respiratory disease in calves treated with Provita Protect (Table 1)
- Provita Protect was at least as effective as antibiotics in scouring calves (together with electrolytes)
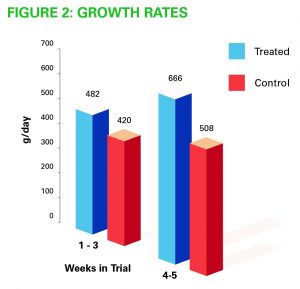
- Calves were heavier at weaning once they had received Provita Protect (Fig 2)
- Over 400 calves were used in various field trials and the aggregate weights of Provita treated calves showed an almost 10 per cent liveweight gain increase over untreated controls
- Fed prophylactically from birth to young calves, scour incidence was less in the Provita Protect treated calves and this was statistically significant.
- At 14 days post treatment in newborn calves, scour incidence was three per cent in Provita treated calves and 12.5 per cent in untreated control calves.
- The trial showed that calves treated with Provita on arrival performed better and were healthier than control calves. Incidence of scour was significantly lower in treated calves at 14 days (P <0.05). A total of 149 calves were used in this trial.
- 52.5 per cent of control calves remained healthy throughout the trial compared with 89 per cent of the Provita Protect treated calves
- Control calves showed an average of 17.88 kg growth over the five-week rearing period, whereas Provita treated calves gained 19.67kg and had less disease, incurring less cost on treatments and antibiotics.
- Laboratory tests and analysis showed Provita Protect to retain adequate counts of live viable organisms for at least five months. If containers are sealed and kept at 2-8C, the shelf life has been shown to be 18 months.
- A significant decrease in pneumonia was evidenced also in Provita treated calves. Seventeen per cent of control calves were treated for respiratory disease compared to five per cent for the Provita-treated calves.
- Incidence of scour plus respiratory disease was 49 per cent in control untreated calves and 11-20 per cent in Provita Protect treated calves
Recent Research on Probiotics and the Gut Microbiome
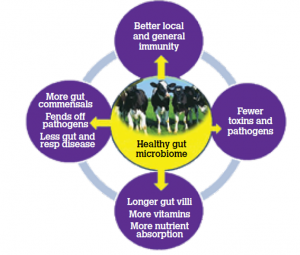
The optimised gut microbiomes now known to be the director not only of gut health but also of general immunity in the body. Medical research shows the beneficial effects of quality probiotics on commensals of the gut and an augmentation of the signalling from the gut to the immune system via SCFA (short chain fatty acids) and other signalling inducers. “Cross talk” exists between the gut and the lung in terms of the optimised gut microbiome facilitating not just local immunity in the gut but also protection in the respiratory system the brain and other body compartments. Eubiosis in the gut (i.e. healthy gut flora) is associated with high SCFA whereas dysbiosis in the gut (pathogenic organisms) is associated with a decrease in SCFA. This recent scientific evidence clearly underpins why in the Provita Protect field trials, the treated calves displayed not only a lower incidence of scour but also a lower incidence of respiratory disease. (Refs 2—6) (Fig 3)
This dual effect has also been noted by customers in the field as shown previously in table 1. The proven effectiveness of Provita Protect in protecting calves from scour and pneumonia, and the resultant heavier weight gains, and improved all-round health status of the treated calves were documented in the Provita Protect Clinical Expert Report ( Barragry 1997, Ref 7).
Written by Dr Tom Barragry PhD, MSc, MVB, MRCVS, Dip ECVPT.
For more information see www.provita.co.uk or email George Shaw, MPharm MPSNI, Technical Adviser, george.shaw@provita.co.uk
References
- Binda S., et al., (2020) Criteria to Qualify Microorganisms as “Probiotic” in Foods and Dietary Supplements. Front. Microbiol. 11:1662. doi: 10.3389/fmicb.2020.01662
- Marsland B., et al., (2018)The Gut-Lung Axis in Respiratory Disease. Scientific Reports. 8:13189
- Enaud R., et al. (2020) The Gut-Lung Axis in Health and Respiratory Disease: A Place for Inter Organ and Inter Kingdom Crosstalks. Frontiers in Cellular and Infection Microbiology. 10:9
- Zhang D., et al., (2020) The Cross-Talk Between Gut Microbiota and Lungs in Common Lung Diseases. Frontiers in Microbiology. 11:301
- Correa-Oliveira R., et al., (2016) Regulation of Immune Cell Function by Short-Chain Fatty Acids., Clinical & Translational Immunology 5:4 e73
- Arpaia N., et al. (2013) Metabolites Produced by Commensal Bacteria promote peripheral regulatory T-cell generation. Nature. 504:451-5
- Provita Protect Clinical Expert Report 1997. Barragry TB.






