Tackling Leptospirosis in the NI swine herd

Research undertaken locally by vet and pig production specialist, Jesus Barobia-Belsue has indicated the prevalence of Leptospirosis in many pig units across the country. Here, Jesus warns that biosecurity protocols and hygiene management are just as important in controlling the disease as vaccination and medication…
Leptopirosis is presumed to be the most widespread zoonosis worldwide (Adler et al. 2010). It is the cause of reproductive loss in swine breeding herds and has been reported in swine from all parts of the world (Ellis 2012).
Leptospirosis is caused by a variety of morphologically similar, but antigenically and genetically distinct, small, motile, aerobic spirochetes belonging to the genus Leptospira. They are thin, helical, motile, gram-negative 6 – 20 µm long organisms, which are often hooked at one or both ends (Ellis 2012).
Twenty-three species have been described to date, including pathogenic and free-living species and certain species that include opportunistic pathogenic strains with more than 250 serovars in 24 serogroups (García Peña and Fraile 2017).
The most pathogenic species found in pigs are Leptospira interrogans and Leptospira borgpetersenii (Ellis 2012).
Pigs can act as maintenance hosts for serovars belonging to the Pomona and Australis serogroup (Bolin 1994, Adler 2015). These serovars are said to be adapted to the pig as a host. Other serovars are accidental by contact with other animal species. The most commonly identified incidental serogroup infections detected in swine are Icterohaemorrhagiae, Grippotyphosa and Tarassovi (Ellis 2012).
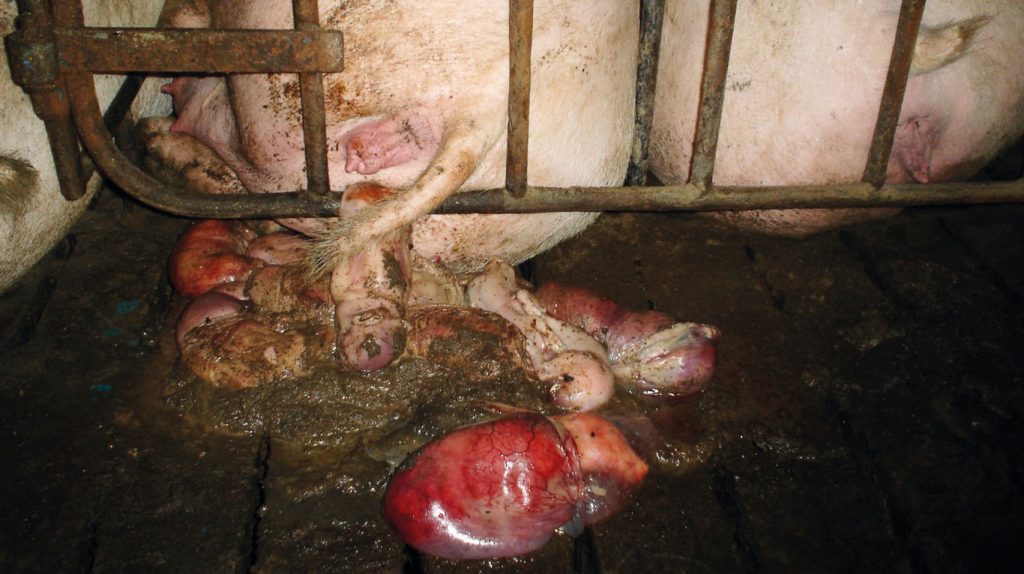
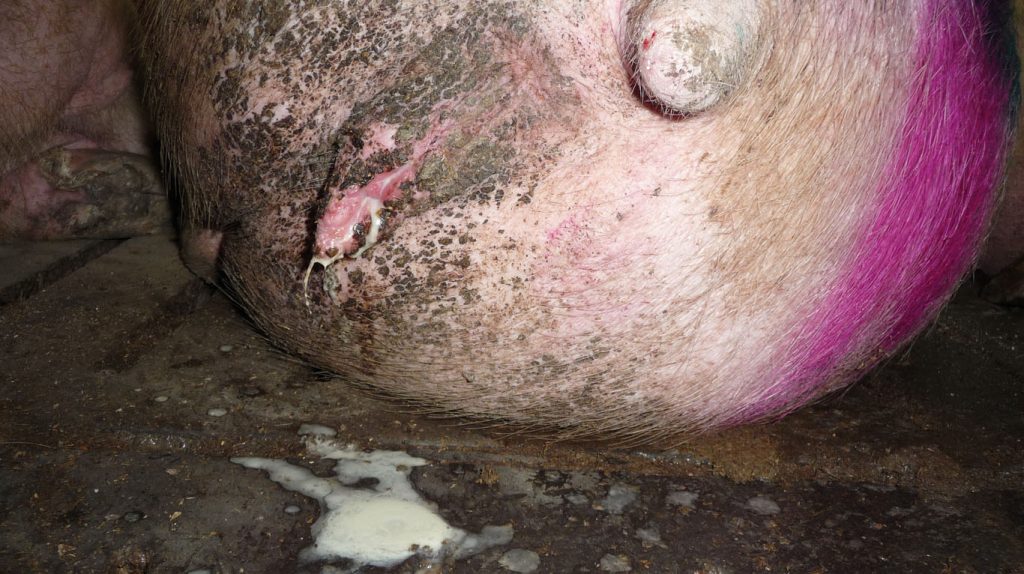
The main sources of infection are urine, placenta, aborted foetuses and vaginal discharge from infected animals, as well as water, grazing and land material contaminated with infected fluids (figure 1 and 2). Infection can take place through damaged skin. A few serovars can be transmitted by the venereal route.
Endemic infections in swine herds generally remain subclinical. However, when a susceptible breeding herd is infected for the first time or its immunity is compromised, considerable losses can occur due to abortion, stillbirths, weak piglets or infertility. Leptospires persist in the kidneys and genital tract of carrier swine and are excreted in urine and genital fluids (Ellis 2012).
Serological testing is the laboratory procedure most frequently used to confirm the clinical diagnosis, to determine herd prevalence and to conduct epidemiological studies. The standard serological test is the microscopic agglutination test (MAT). A titre of 100 is taken as positive for the purpose of international trade (OIE 2014). The MAT is useful in diagnosing acute infection. However, it has severe limitations in the diagnostic of chronic infection in individual pigs. Infected animals may have MAT titres below 1:100 (Ellis and others 1986a,b).
There are very few recent studies of the prevalence of Leptospirosis in pigs worldwide. The seroprevalence of an study in Colombia was 55.9 per cent (Calderón and others 2013); 3.5 per cent seroprevalence from 137 slaughtered pigs in Brazil (Miraglia and others 2008); 1.32 per cent to 2.68 per cent seroprevalence in pigs in Poland from 2011 to 2015 (Zebek and others 2016); 1.2 per cent seroprevalence in pigs in West Germany (Schonberg and others 1984); 73.3 per cent seroprevalence in sows in the Mekong Delta, Vietnam (Boqvist and others 2002); 8.17 per cent seroprevalence in fattening pigs from five provinces in Vietnam (Suk Lee and others 2017); and, 20.2 per cent seroprevalence in swine in Germany from 2011 to 2016 (Strutzberg-Minder and others 2018).
There are no seroprevalence studies in pigs in the U.K., Ireland or Northern Ireland.
Materials and methods
Eighteen pig farms with fertility problems in Northern Ireland were tested for antibodies against different Leptospira serovars in 2018 using the MAT method. The serovars tested were Australis (AUS), Bratislava (BRA), Autumnalis (AUT), Canicola (CAN), Grippotyphosa (GRI), Copenhageni (COP), Icterohaemorrhagiae (ICT), Pomona (POM), Altodouro (ALT), Hardjo (HAR), Saxkoebing (SAX) and, Tarassovi (TAR).
Serovars from the same serogroup can cross-react leading to the wrong reading of serovar affected. Serovars AUS and BRA can cross-react because they are from the same serogroup (Australis). Serovars COP and ICT can cross-react because they are from the same serogroup (Icterohaemorrhagiae). Serovars POM and ALT can cross-react because they are from the same serogroup (Pomona). Serovars HAR and SAX can cross-react because are from the same serogroup (Sejroe).
Only sows with fertility problems were tested.
Results
Fifteen pig farms had samples with titres of ≥1:100 to one or more Leptospira serovars (Table 1).
There were a total of 137 blood samples taken. Forty-four (32.1 per cent) were positive to Leptospirosis and 93 samples (67.9 per cent) were negative.
Table 2 shows the frequency of the serovars detected in this study.
Prevalence
The results of the present study indicate that Leptospira is present in many pig units. The finding of this spirochete by MAT could potentially play a key role in the fertility performance in the herd (Ellis 2012). Other factors could also alter fertility performance in pig units. These may include other infections pathogens (e.g. PRRS -Porcine Reproductive and Respiratory Syndrome-, SIV -Swine Influenza Virus-, porcine parvovirus, erysipelas, endometritis, pyelonephritis), management practices and seasonal and/or nutritional imbalances.
The main serovars detected in these sows were BRA (81.8 per cent) followed by AUS (68.2 per cent), ICT (38.6 per cent), COP (34.1 per cent), POM (34.1 per cent), HARD (27.3 per cent), GRI (22.7 per cent). These results are similar to the surveillance of Leptospira infection in swine in Germany carried out by Strutzberg-Minder (2018).
The finding of serovars belonging to the Australis serogroup as the most frequent confirms Bolien (1994) and Adler (2015) findings.
Serovars from the serogroup Icterohaemorrhagiae were detected at high levels of frequency after the Australis serogroup. This is an incidental non-pig specific serogroup. Rodents are the main maintenance host to the ICT and COP serovars (Ellis 2012). The finding of these serovars in pig units indicates a high level of exposure to rodents in the farm.
Ellis (2012) states that Serovar HAR infection is maintained by cattle worldwide, and where cattle and pigs come in close contact, the opportunity arises for infection in pigs to occur, as seen in the farms seropositive in this study.
Little is known of the epidemiology of serovar CAN infection in pigs. The recognised maintenance host for this serovar is the dog (Ellis 2012). It is important that farm dogs are sufficiently vaccinated and that they don’t come into contact with livestock.
Control
The traditional approach for controlling Leptospirosis in N.I. and Europe was medication in-feed for several weeks at high levels of tetracyclines or sulfadiazines combined with managerial and hygienic measures such as vermin control and washing-disinfection-drying techniques. This measure is unpopular due to current demand for responsible use of antibiotics in food-producing animals in the U.K and the E.U.
The importation of commercial vaccines available in other continents was taken into consideration nine years ago. This action helped to reduce the use of antimicrobials in livestock without negatively affecting the reproductive performance. Recently, new Leptospira swine vaccines have been launched and are at the process of being launched in the U.K. and the E.U.
It is important to conclude that vaccination and/or medication are not the only measures to take into consideration. Biosecurity, management and hygiene must be incorporated into the Leptospira treatment plan combined with proper diagnostic tools.
Jesús Borobia-Belsué MSc, CertPM, MRCVS is a veterinary surgeon currently practising in Northern Ireland. He graduated with a degree in veterinary medicine in Barcelona (Spain) in 1998 before obtaining a Master of Science in pig production from Aberdeen University in 1999. In 2007, he gained the Certificate in Pig Medicine from the Royal College of Veterinary Surgeons. He is the senior pig vet at Moss Veterinary Ltd. with clients throughout the island of Ireland. He has published a number of articles in peer-review magazines and presented at both national and international pig events.






